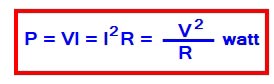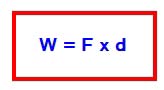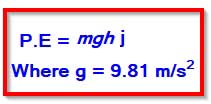Fundamental OF Electrical Engineering | Work_Energy_Power| Current and Voltage Symbol
The measurement of a quantity involves a unit and a number. The unit is a reference amount of the quantity, and the number expresses the ratio of the magnitude of that quantity to the magnitude of the unit. When the unit of a quantity is assigned an arbitrary value, that unit is called a fundamental unit and the quantity a fundamental quantity.
The International System of Units referred to as the SI system is almost universally used for scientific and engineering purposes. The S.I. the system is divided into seven fundamental units and two supplementary units. The seven fundamental or base units are length, mass, time, Electric current, temperature, amount of substance, and luminous intensity. The two supplementary units are the plane angle and solid angle.
[ninja_tables id=26963]
The other units can be derived from the Fundamental units and Supplementary units. These derived units are classified into three main part
- Electrical Unit
- Heat Unit
- Mechanical unit
Electrical Unit
Electrical Work:
In an electric circuit or a system, work is said to be done when a charge is transferred from one point to other through a potential difference. The SI unit of Work is Joule.So If V is the potential Difference in volts Q is the charge in coulombs then we can write
Work Done = Charge × Potential Difference
W = Q × V
★The work done is said to be One joule if the charge of one coloumb is moved through the potential difference of one volt.
Electrical Power
The rate at which the electric work is done in an electric circuit is called an electric power. The SI unit of power is joule per second or watt.
★The Power consumed in an electric circuit is one watt If the potential difference of 1 volt is applied is applied across the circuit that causes 1 ampere current to flow in the circuit.
According to Ohm’s law
V = IR and I = V/R
Therefore the Power can be expressed as
Electrical Energy
Electric energy is the total amount of electrical work done in an electrical circuit. The electric energy can also be defined as the product of power and time. The S.I Unit of Electrical- Energy is joule or watt-sec.
★The energy consumed by the circuit is said to be 1 joule or watt-sec when it utilize power of 1 watt for 1 second.
The Energy is also expressed in kWh and 1 kWh is equal to 1 unit.This Unit (kWh) is also called as Board of trade Unit.
1 kWh = 1000 watt-hour
= 1000 x 3600 = 36 × 105 watt-second or joules.
Mechanical Unit
Work
Work is said to be done in the body when a force acts on it and the body moves through some distance.The work is Measured in joule.
Suppose a body moves through the distance d under the force F then
★The work done is 1 joule if a force of 1 newton moves the body through one meter in the direction of force.
Power
It is defined as the rate of work done with respect to time. The SI unit of power is Joule/Second or watt.
★The power dissipated is one watt if one joule of energy is expended in One second.
Energy Expended = Power x Time = Work Done
Energy
It is defined as the capacity to do work. The work is done always at the cost of energy and the work done is stored in the body in the form of energy. Hence work and energy are measured in the same unit i.e Joule.
The mechanical energy is divided into two parts
- Kinetic Energy
- Potential Energy
Kinetic Energy
It is defined as the energy possessed by the body due to its motion. If the body of the mass m moving with the velocity V then the kinetic energy is
Potential Energy
It is defined as the energy possessed by the body due to its position. When a body of mass m is lifted vertically through height of h then the potential energy is
P.E = mass × gravitational force × height
Heat and Energy in a Thermal System
When a body is heated from the temperature t1 to t2, heat is gained by the body. When the body is cooled from the temperature t2 to t1, heat is lost by the body.
The amount of heat gained or lost by a body is different in different conditions and depends on the following factors:
- The amount of heat gained or lost by a body is directly proportional to its mass (m).
- The amount of heat gained or lost depends upon the nature of the material of the body.
- The amount of heat gained or lost by a body depends upon the change in its temperature (t1 – t2).
The units of heat are calorie and kilocalorie.
★One calorie is the amount of heat required to raise the temperature of 1 g of water by 1°C.
1 kilocalorie = 1000 calories
Since heat is a form of energy, it can also be measured in joules.The relation between joule and other units of heat is as given below.
1 calorie = 4.186 joules
1 Kilocalorie = 4186 joules
1 joule = 0.2389 calories
Specific Heat Capacity
The amount of heat required to change the temperature of 1 kilogram of the substance through 1-degree Kelvin is called as specific heat of the substance. The unit of specific heat energy is J/KgºC or J/Kg K.
Sensible Heat
The quantity of heat loss or gained or when the change in temperature occurs is called as the sensible heat. This can be calculated as
Sensible heat = mCΔt
m = mass of substance in Kg
C = specific heat in J/Kg – °K
Δt = t2 – t1 change in temperature
Latent heat
The quantity of heat required to change the state of the substance i.e from solid to liquid to gas without the change in the temperature is called as latent heat. During the conversion, the temperature of the body remains constant at its melting point.
Latent Heat = m × L Joules
m = mass of substance in Kg
L = specific latent heat or specific enthalpy
The unit of specific latent heat is J/Kg while the unit of latent heat is Joules.
Total heat = Sensible heat + Latent Heat
[ninja_tables id=26985]
The magnitude or the size of the Electrical measurement may result in an extremely large number for the specified unit. For e.g the value of current can be lower than 0.000001 A or it can be higher than 100000 A. Therefore multiple and the submultiple of a unit are used to eliminate the impracticable aggregate of the numbers.
The SI system is based on multiple of 10 i.e multiple and submultiple of each unit are used when describing the magnitude of measurement.
[ninja_tables id=26989]
The Capital letters are used for the abbreviations of Multiples while the small letters are used for the abbreviations of Submultiples. The exception is kilo for which the abbreviation is the lower case k, not the capital K.
★The Prefixes centi, deci, Deka, and hekto are not used with electrical quantities. The remaining prefix progress with power of 10 and are divisible by three.
Current and Voltage Symbol
- Capital letter with capital subscript denotes DC or average quantity. Example: VA.
- Lowercase letters with lowercase subscript denotes a small signal of zero average value. Example: ia.
- Capital letter with lowercase subscript denotes the RMS value of an alternating quantity, its Fourier transform, or its Laplace transform. Example Vi.
- Lowercase letters with capital subscript denotes a total instantaneous quantity. Example iB.
- The double subscript in a current symbol denotes a current flowing from the terminal designated by the first subscript to the terminal designated by the second subscript. Example: iab.
- The double subscript in a voltage symbol denotes a voltage drop from the terminal designated by the first subscript to the terminal designated by the second subscript. Example: vab.